Subsequent addition of GlcNAc to lipid I by MurG results in inward-oriented lipid II molecules. The enzyme MraY couples the UDP-MurNAc-pentapeptide to undecaprenyl phosphate to produce the membrane-anchored lipid I ( Manat et al., 2014). The precursors for PG biosynthesis, uridine diphosphate- N-acetylglucosamine (UDP-GlcNAc) and UDP-MurNAc-pentapeptide, are synthesized in the cytoplasm by the enzymes MurA-F ( Barreteau et al., 2008). The few variations are either a change in the amino acid sequence of the stem peptide (almost always the di-amino acid at position 3) or the consequence of accessory reactions that modify the basic subunit (e.g., O-acetylation of sugars or amidation of dicarboxylic amino acids) ( Vollmer, 2008 Cava and de Pedro, 2014). The structure of the monomeric subunit, N-acetyl-glucosaminyl- N-acetyl-muramyl-L-alanyl-D-glutaminyl-L-(meso)diaminopimelyl-D-alanyl-D-alanine (GlcNAc-MurNAc-L-Ala-D-Glu-L-mesoDAP-D-Ala-D-Ala), is remarkably conserved throughout the bacterial phylogenetic tree. PG is a polymer of glycan chains crosslinked by peptides. PG is present in essentially all bacteria and wraps the cytoplasmic membrane like an elastic net ( de Pedro and Cava, 2015). Therefore, morphogenesis should be viewed as a major evolutionary and adaptive process that contributes greatly to prokaryotic ubiquity and versatility.īacterial shape is primarily dictated by the peptidoglycan (PG) sacculus ( Salton and Horne, 1951 Weidel et al., 1960), a polymeric macromolecular structure that surrounds the cytoplasmic membrane and is the only “solid” element in the bacterial envelope. Shape dictates the interactions between a bacterial cell and its environment, most notably small-molecule traffic (via the surface/volume ratio), motility, formation of multicellular aggregates, habitat colonization (including eukaryotic hosts and consequently pathogenesis and symbiosis), predation, and resistance (see Young, 2006, for a comprehensive review). This is evidenced by the development of aberrant morphology upon mutations. To ensure constant bacterial morphology over generations despite these forces, shape maintenance must be an active process guided by robust regulatory circuits. Bacterial shape is genetically determined, but physical forces (internal and external) exerted on cells are increasingly recognized as major players in morphogenesis. The characteristic morphology of a bacterial species is maintained through countless generations but is periodically modified within set limits during bacterial division and life cycles (Figure 1). 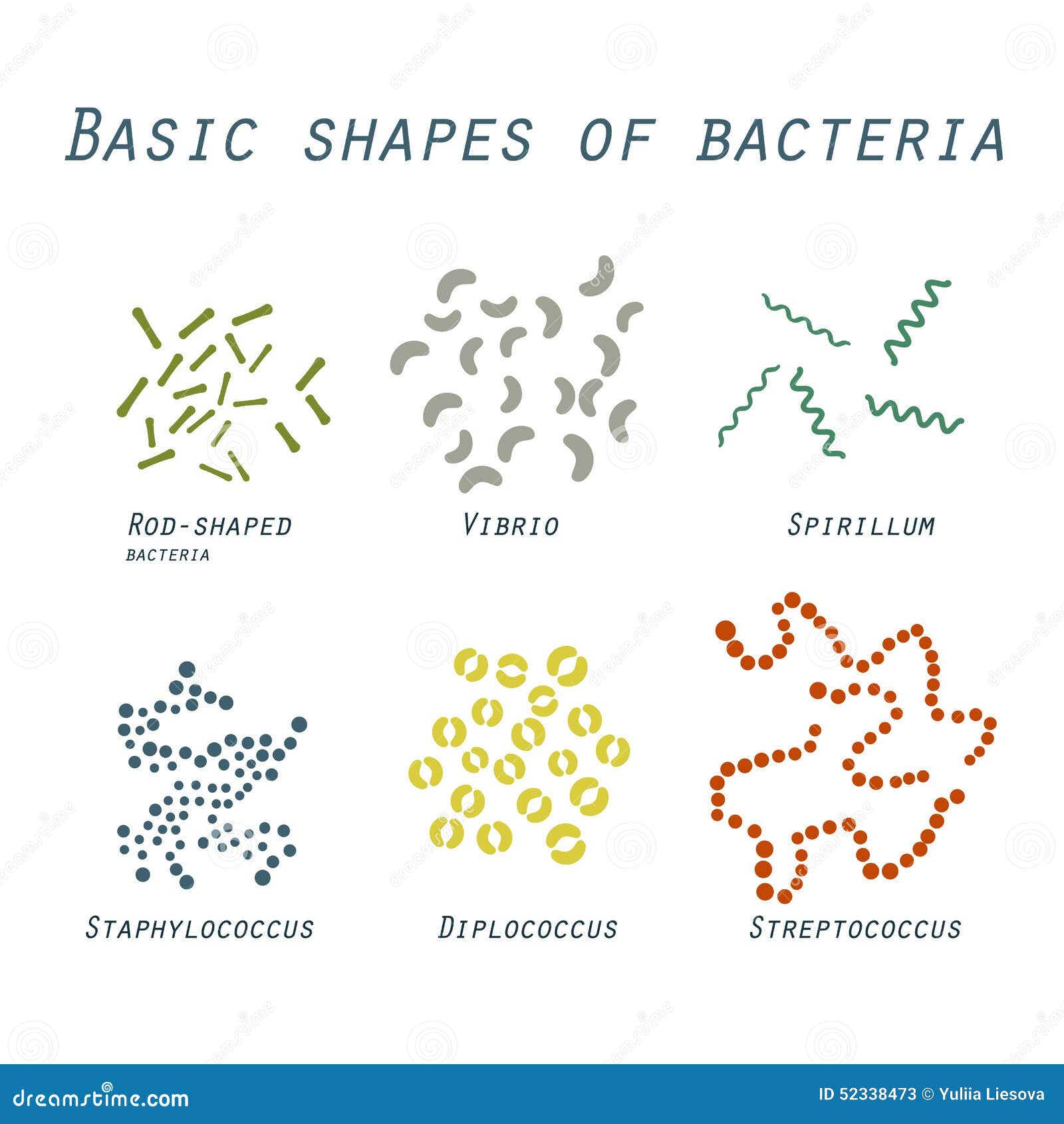
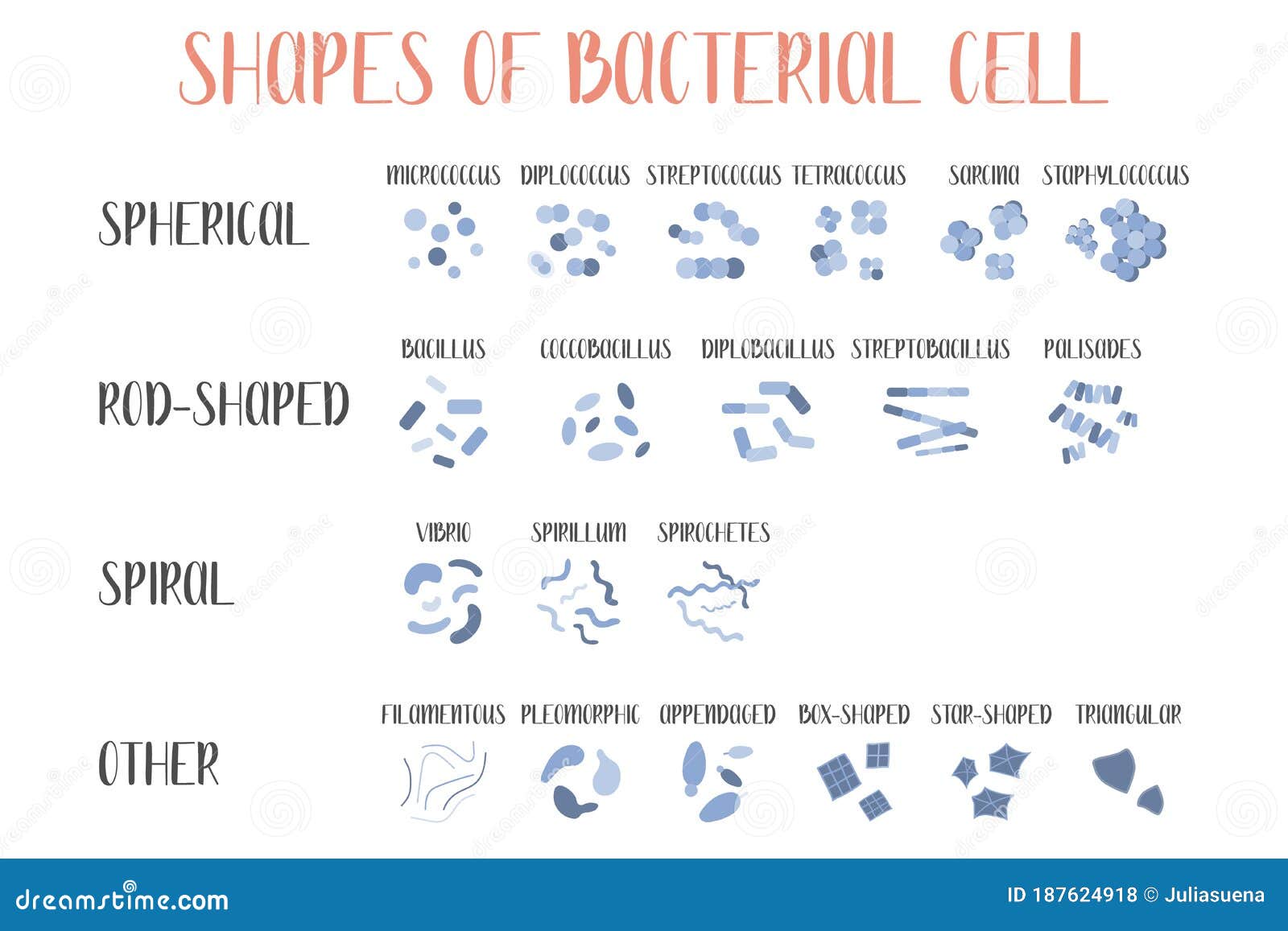
In addition to the well-known rods and cocci, more exotic shapes such as stars, mustaches, serpentines, and branches represent a large, although undefined, proportion ( Young, 2006 Kysela et al., 2016). The variation of bacterial cell shapes is often underappreciated. Here, we review the molecular determinants underlying morphology, discuss the evidence linking bacterial morphology to niche adaptation and pathogenesis, and examine the potential of morphological determinants as antimicrobial targets. Accumulating evidence supports critical roles of morphogenetic processes in bacteria-host interactions, including pathogenesis. 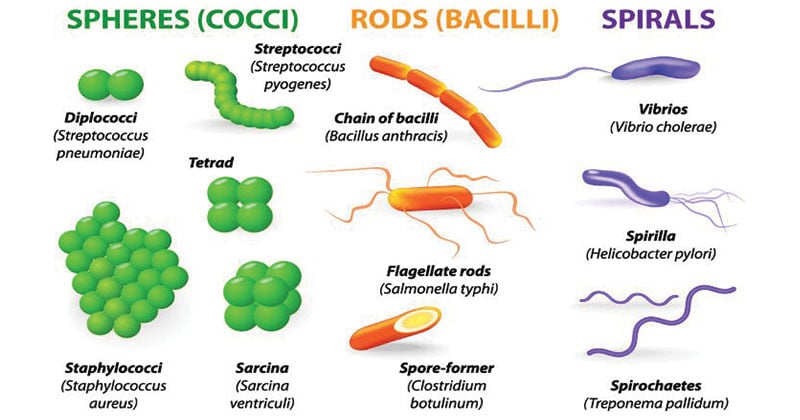
Some morphological determinants act as a cytoskeleton to guide biosynthetic complexes spatiotemporally, whereas others modify the PG sacculus after biosynthesis. The species-specific shape of the PG sacculus at any time in the cell cycle is the product of multiple determinants. Bacterial morphology is ultimately dictated by the net-like peptidoglycan (PG) sacculus. 
Although the characteristic shape of a bacterial species remains unchanged for vast numbers of generations, periodical variations occur throughout the cell (division) and life cycles, and these variations can be influenced by environmental conditions. Shape affects critical biological functions, including nutrient acquisition, motility, dispersion, stress resistance and interactions with other organisms. Specific shapes are the consequence of adaptive pressures optimizing bacterial fitness. 2Centro de Biología Molecular “Severo Ochoa” – Consejo Superior de Investigaciones Científicas, Universidad Autónoma de Madrid, Madrid, Spainīacterial morphology is extremely diverse.1Laboratory for Molecular Infection Medicine Sweden, Department of Molecular Biology, Umeå Centre for Microbial Research, Umeå University, Umeå, Sweden. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
